CARTs IN LYMPHOMA AND MULTIPLE MYELOMA
CART-cell therapy targeting CD19 has transformed the treatment landscape for relapsed or refractory (R/R) B-cell lymphomas.
Unprecedented response rates and durable outcomes in high-risk B-cell lymphoma patients led to the rapid FDA approval of anti-CD19 CAR T-cell therapy for various lymphoma subtypes making it standard of care for patients with R/R aggressive non-Hodgkin’s NHL after two or more lines of therapy.
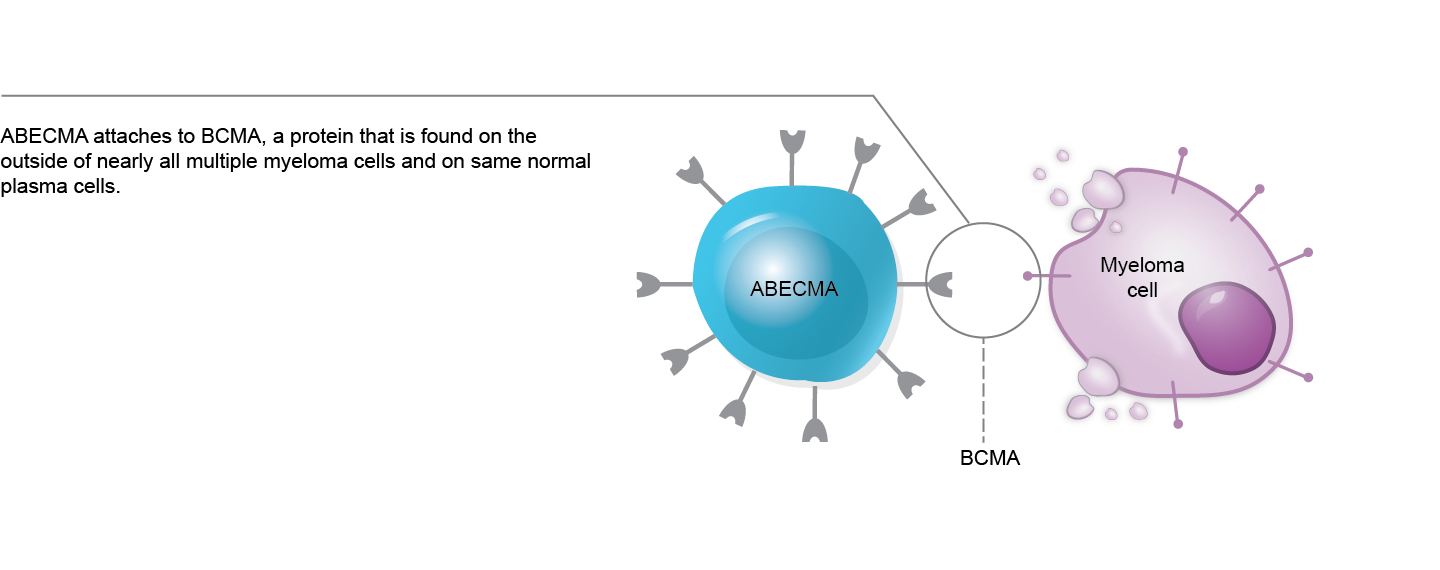
The BCMA expression is constricted to the mature B-lymphoid compartment, making it a promising target in patients with MM. The US FDA has approved two BCMA-targeting CAR T-cell therapies for MM: Ide-cel and Cilta-cel.
BiTEs IN LYMPHOMA AND MULTIPLE MYELOMA
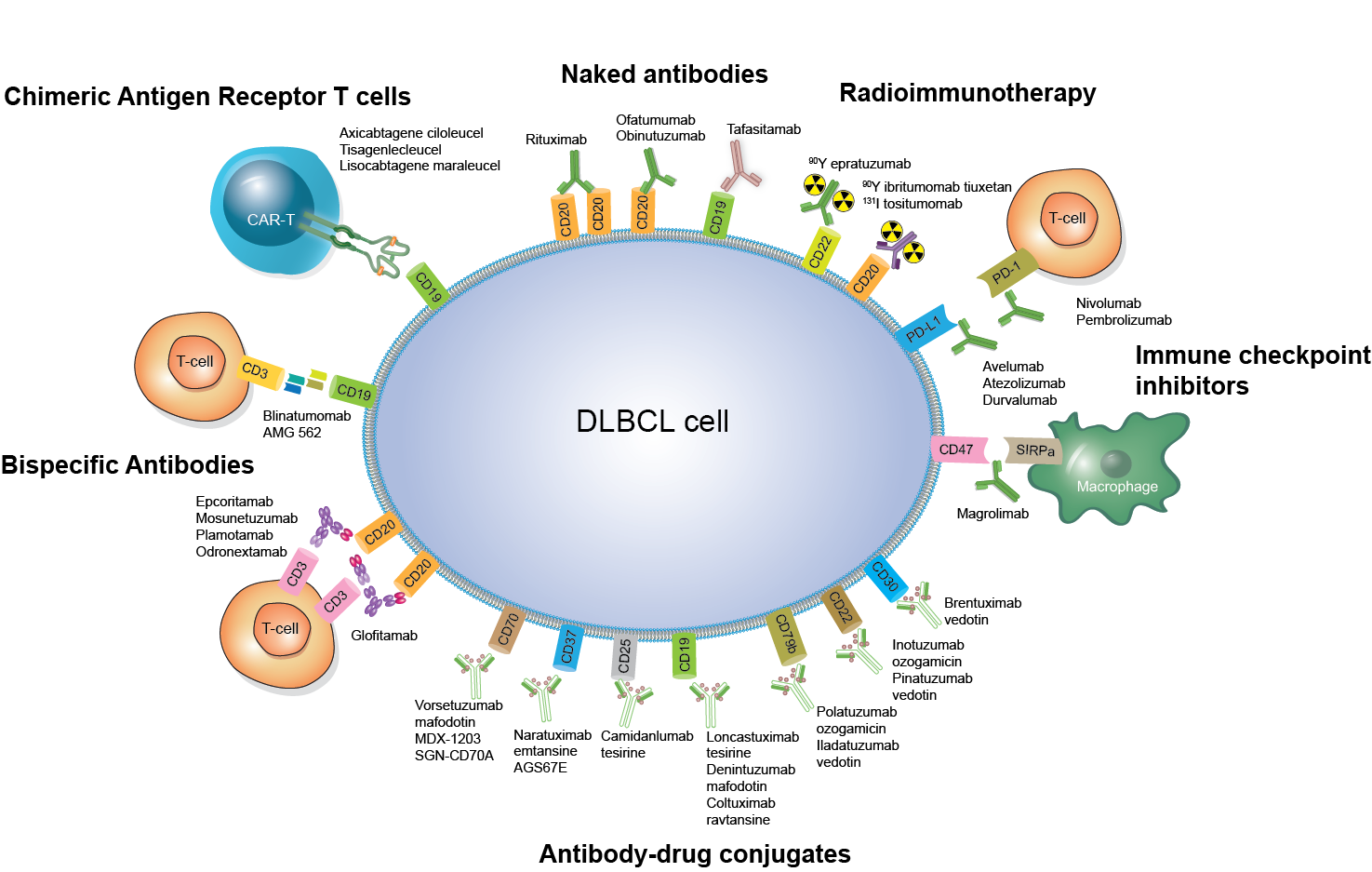
Anti-CD20xCD3 BsAbs have shown remarkable single-agent efficacy in heavily pretreated B-NHL patients, with a manageable toxicity profile predominantly characterized by T-cell overactivation syndromes.
Two BsAbs, epcoritamab and glofitamab, have recently received FDA approval for the management of relapsed/refractory (R/R) DLBCL.
The most advanced CD20-directed T-cell engagers in B-NHL include mosunetuzumab, glofitamab, epcoritamab, and plamotomab.
ADCs IN LYMPHOMA & LEUKEMIA
In recent years, three ADCs – brentuximab vedotin, polatuzumab vedotin, and loncastuximab tesirine – have been FDA-approved and are establishing their roles in lymphoma treatment.
Advances in ADC design have enhanced their efficacy and safety, driving their integration into clinical practice. A growing number of novel ADCs are generating significant interest as promising treatment options for lymphoma.
FLT3 INHIBITORS IN AML
Since the discovery of FLT3 mutations and their prognostic significance, several FLT3-targeted therapies have been developed. Midostaurin, approved in the U.S. and Europe, is used with induction and consolidation chemotherapy for newly diagnosed FLT3-mutated AML, while Gilteritinib is approved as monotherapy for relapsed or refractory FLT3-mutated AML.
OTHER TARGETED MOLECULAR THERAPIES IN AML
The pathological interactions between menin and KMT2A in KMT2Ar leukemias and NPM1mut AML has led to the recognition of a new promising target for therapies in this subset of leukemias.
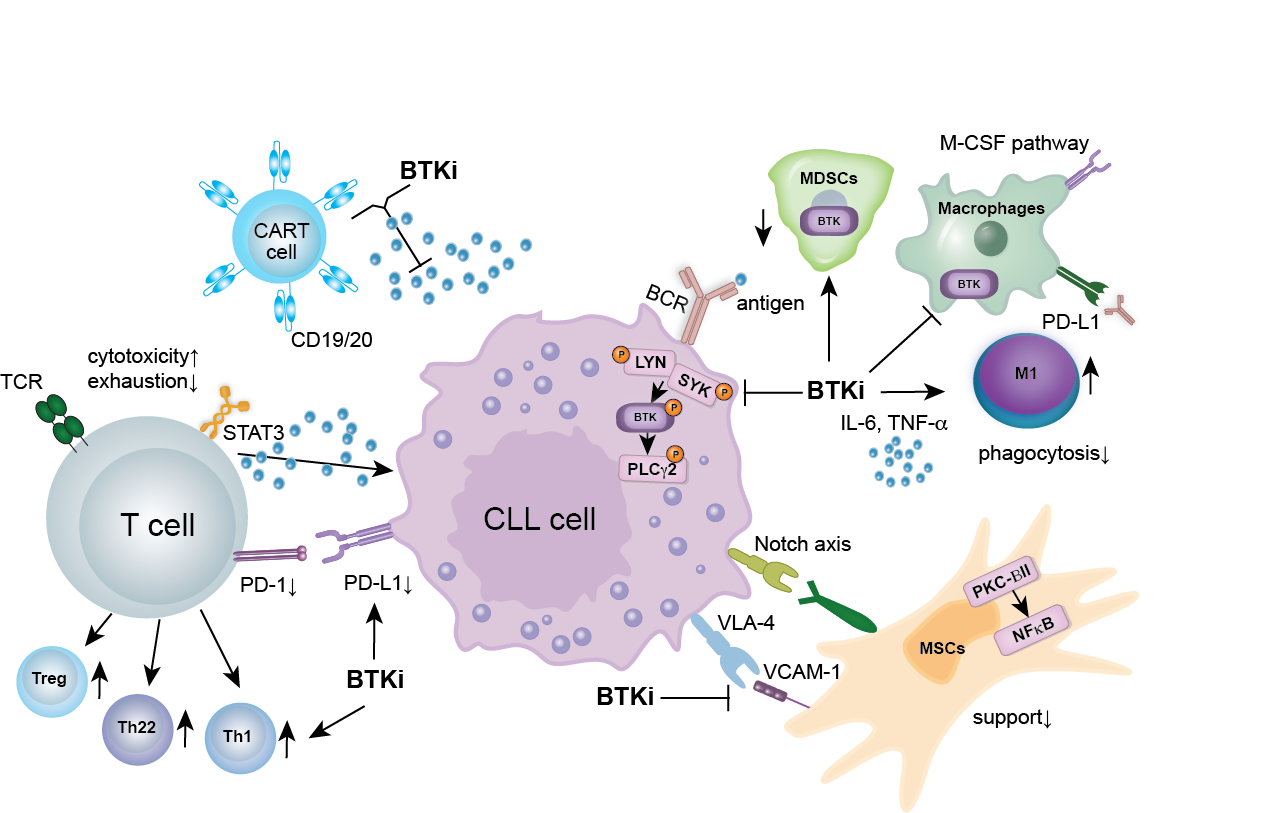
BTK AND BCL-2 INHIBITORS IN CLL
BTKi are the standard of care in CLL, MCL, WM, and MZL and have recently been approved for use in Europe in relapsed FL, in combination with Obinutuzumab. The extensive clinical use of BTKi has revealed challenges such as cardiovascular and bleeding side effects, as well as resistance mutations. In response, new classes of “second-generation” and “reversible” BTK inhibitors have been developed to address these issues.